Requirements Management For Regulated Products
Supporting innovators through the process of Product Realisation and ensuring compliance with industry standards

Navigating Complexity
Clear technical requirements are key to building successful products (as well as regulatory compliance), but can be a confusing mixture of jargon and complex processes.
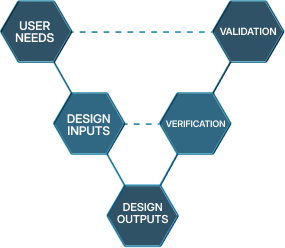
Requirements Management
Upstart Bio helps teams establish Requirements Management processes to turn market data and ideas into technical specifications that can be effectively Verified and Validated. We take a hands-on approach that uses industry-specific experience but also promoting effective collaboration and communication across different functions within organisations.
Experienced Support And Resources
Helping team develop reliable, compliant products that meet user needs.
Clarity
Clear, right-sized advice for start-ups.
Experience
Knowledgeable support and practical advice backed by over 25 years in the Life Science sector.

Quality And Compliance
Practical guidance through complex processes.

